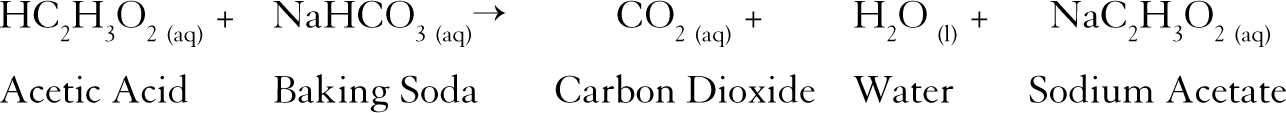
The second largest use of sodium bicarbonate is as an additive in animal feed. The acidity provides a sharp taste and helps to preserve a food. Baking soda is also used as an additive in foods and drinks to provide effervescence (a bubbling, fizzing, or sparkling effect) or to maintain an acidic environment in the food. Baking powder is a more efficient leavening agent in baking than is baking soda by itself. One of the most common compounds mixed with sodium bicarbonate in baking powder is tartaric acid (HOOC(CHOH) 2COOH), or its salt, potassium bitartrate (HOOC(CHOH) 2COOK). Baking powder differs from baking soda in that it includes an acidic compound that reacts with sodium bicarbonate to produce carbon dioxide. About one-third of that amount was used by the food products industry, primarily in the manufacture of baking soda (pure sodium bicarbonate) and baking powder (a mixture of sodium bicarbonate and at least one other compound). 
In the 1980s, restorers used an aqueous solution of the compound to clean the Statue of Liberty.Īn estimated 560,000 metric tons (615,000 short tons) of sodium bicarbonate were consumed in the United States in 2003. Sodium bicarbonate is a very effective cleaning agent for certain materials.Although sodium carbonate is the desired product in this reaction, sodium bicarbonate can also be obtained by deleting the final step by which it is converted into sodium carbonate. The sodium bicarbonate is then heated to obtain sodium carbonate. Solvay developed a procedure by which sodium chloride is treated with carbon dioxide and ammonia, resulting in the formation of sodium bicarbonate and ammonium bicarbonate. Sodium carbonate had long been a very important industrial chemical for which no relatively inexpensive method of preparation existed. The Solvay process was invented in the late 1850s by Belgian chemist Ernest Solvay (1838–1922) primarily as a way of making sodium carbonate. Sodium bicarbonate is also obtained as a byproduct of the Solvay process. Since the bicarbonate is less soluble than the carbonate, it precipitates out of solution and can be removed by filtration. In the first method, carbon dioxide gas is passed through an aqueous solution of sodium carbonate (Na 2CO 3): 
Sodium bicarbonate is made commercially by one of two methods. Consumers use the product for cooking, cleaning, and deodorizing homes. The company still produces 90 percent of all the baking soda used for household purposes in the United States. The Church-Dwight operation grew over the years to become the largest producer of household baking soda, now sold under the name of Arm & Hammer® baking soda. They started their company in the kitchen of Dwight's home, making the product by hand and packing it in paper bags for sale to neighbors. In 1846, Connecticut physician Austin Church (1799–?) and John Dwight (1819–?) of Dedham, Massachusetts, founded a company to make and sell sodium bicarbonate. Since all the compounds present in this reaction are safe for human consumption, sodium bicarbonate makes an ideal leavening agent.Ĭommercial production of sodium bicarbonate as baking soda dates to the late 1700s. Sodium bicarbonate produces this effect because, when heated or dissolved in water, it breaks down to produce carbon dioxide (CO 2) gas: 
A leavening agent is a substance that causes dough or batter to rise. For centuries, people around the world have used sodium bicarbonate as a leavening agent for baking. Ancient Egyptian documents mention the use of a sodium bicarbonate and sodium chloride solution in the mummification of the dead. Sodium bicarbonate has been used by humans for thousands of years. Soluble in water insoluble in ethyl alcohol Sodium, hydrogen, carbon, oxygen COMPOUND TYPE:Ībout 50☌ (120☏) decomposes BOILING POINT: KEY FACTS OTHER NAMES:īicarbonate of soda baking soda FORMULA: The compound's primary uses are as an additive in human and animal food products. Sodium bicarbonate (SO-dee-um bye-KAR-bun-ate) is a white, odorless, crystalline solid or powder that is stable in dry air, but that slowly decomposes in moist air to form sodium carbonate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
